Original Papers
I try to introduce the essence of my work in easier words than in the manuscript, here. If you want to check simple publication list, please visit my Google Scholar page, ORCiD page, or Scopus page (Links).
論文の要点についての説明を入れています。シンプルなリストをご覧になりたい方は、Google Scholar、ORCiD、または Scopus のページを覗いてくださると幸いです (Links)。
2021
36. Controlling Reactivity of Copper(II)-acylperoxide Complexes
Yuma Morimoto; Makito Kawai; Aya Nakanishi; Hideki Sugimoto; Shinobu Itoh
Inorg. Chem. 2021, ASAP (DOI: doi.org/10.1021/acs.inorgchem.1c00475)
35. Hydroxylation of Unactivated C(sp3)–H Bonds with m-CPBA Catalyzed by FeIII-complex Supported by Trianionic Planar Tetradentate Ligand
Yuma Morimoto*; Shinichi Hanada; Ryusuke Kamada; Arisa Fukatsu; Hideki Sugimoto;
Shinobu Itoh*
Inorg. Chem. 2021, ASAP (DOI: 10.1021/acs.inorgchem.0c03469)
共有結合性の極めて高いアルカンのC–H結合は、なかなか切断できないことが知られています。特に、3級や2級のC–H結合と比較すると、分子の末端に位置する1級炭素上のC–H結合は特に切断が難しい結合です。この研究では、フェノールとβ-ジケチミネートからなるトリアニオン性の配位子を持つ鉄錯体を使って、強固なアルカンの酸化を試みました。過酸を酸化剤として利用することで、この錯体触媒は1級炭素を酸化することが分かりました。さらに、濃度を高めることが難しいために反応に利用することが難しいエタンガスを酸化できることも分かりました。触媒反応過程について速度論的な検討を行い、通常鉄系の触媒で生じる活性種である鉄オキシド錯体ではなく、鉄過酸錯体が反応活性種である可能性が示唆されました。
2020
34. Modelling a ‘histidine brace’ motif in mononuclear copper monooxygenases
Arisa Fukatsu; Yuma Morimoto; Hideki Sugimoto; Shinobu Itoh
Chem. Commun. 2020, 56, 5123–5126 (DOI: 10.1039/d0cc01392g)
In nature, there are copper-containing metalloenzymes that can transform with very low chemical reactivity, such as cellulose and methane. The copper centers of these enzymes have a characteristic structure called “histidine brace”, which is supported by a coordination structure consisting of two imidazole residues and an N-terminal amine. The two imidazole ligands trans-coordinated to the copper center, have their π-planes tilted at about 70° to each other. No model complex has ever been known to successfully mimic this structure. Dr. Fukatsu succeeded in synthesizing a copper complex that mimics the active center of such enzymes. This complex was able to carry out the oxidation reaction of alkanes using hydrogen peroxide as an oxidant.
自然界にはセルロースやメタンといった、一般的に酸化することが極めて困難な基質を参加することができる銅含有金属酵素があることが知られています。これらの酵素の銅中心は、ヒスチジンブレイスと呼ばれる、2つのイミダゾール残基ととN-末端アミンから成る特徴的な配位構造にサポートされた構造を持っています。銅中心に対してトランス位から配位子した2つのイミダゾール配位子は、互いのπ平面が70°程度傾いていますが、このような構造を上手に模した銅錯体は、これまで知られていませんでした。深津博士研究員は、このような酵素の活性中心の特徴をうまく模倣した銅錯体の合成に成功しました。この錯体は、過酸化水素を酸化剤としてアルカンの酸化反応を行うことができました。銅錯体のEPR測定および、そのスペクトルの解析を行いました。

33. Controlling Coordination Number of Rhodium(III) Complex by Ligand-Based Redox for Catalytic C–H Amination
Daiki Fujita; Akira Kaga; Hideki Sugimoto*; Yuma Morimoto; Shinobu Itoh*
Bull. Chem. Soc. Jpn. 2020, 93, 279–286 (DOI: 10.1246/bcsj.20190291)
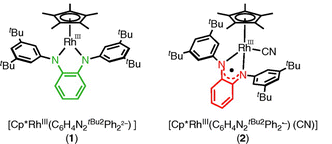
The main finding of this study is a novel method to control the ligand exchange process of Rh complex. As known well, ligand exchange of Rh(III) complex requires high-activation energy mainly due to significant ligand field stabilization energy. The diaminobenzene ligand can stabilize penta-coordinated Rh(III)-center with an efficient π-donation, and one-electron oxidation of the diaminobenzene ligand induces coordination of external ligand to accomplish the octahedral structure. This data suggest that the ligand redox can be utilized as a switch to facilitate ligand exchange on a stubborn Rh(III) complex. This Rh complex shows catalytic activity in amination reaction under relatively moderate temperature conditions.
多くのRh(III)錯体は、 その大きな配位子場安定化エネルギーのために6配位構造を強く好むことが知られています。Rh錯体は高い触媒活性を示すものも多いため興味を持たれていますが、配位子交換の遅さがネックになることもあります。
本研究で合成された、ジアミノベンゼンを2つ目の配位子とするRh(III)Cp*錯体は、珍しい5配位構造を取ることがわかりました。量子化学計算から、ジアミノベンゼン配位子からの強いπ供与がこの5配位錯体を安定化していることが示唆されました。また、この配位子を1電子酸化すると、外からさらにもう1つの配位子を取り込んで6配位錯体になることがわかりました。これはリガンドのレドックスが、頑固なRh(III)錯体の配位子交換を促すスイッチとして利用できる可能性を示しています。この錯体は、比較的温和な温度条件で、アジドを酸化剤としたアミノ化反応活性を示すこともわかりました。DFTとEPRのパートを担当しました。
2019
32. Direct Observation of Primary C–H Bond Oxidation by an Oxido-Iron(IV) Porphyrin π-Radical Cation Complex in a Fluorinated Carbon Solvent
Yuma Morimoto*; Yuki Shimaoka; Yuri Ishimizu; Hiroshi Fujii*; Shinobu Itoh*
Angew. Chem. Int. Ed. 2019, 58, 10863-10866 (DOI: 10.1002/anie.201901608)
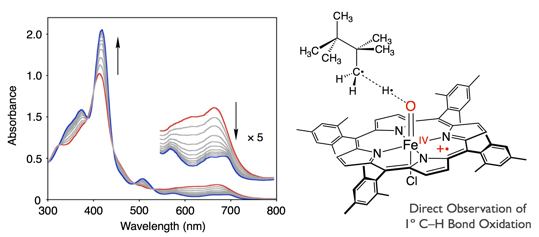
Reactivity studies for oxido-iron(IV) porphyrin π-radical cation species [Fe(IV)(O)(Por·+)] had to be conducted under super-cold conditions (i.e. -60°C) due to high-reactivity of the complex. Such conditions also limit the substrates to highly reactive compounds, such as benzylic positions of alkylbenzene.
In this work, we demonstrated usefulness of trifluorotoluene as a solvent of porphyrin chemistry. By employing the new solvent system in porphyrin chemistry, we could elongate the lifetime of [Fe(IV)(O)(Por·+)] by 700-fold compared to the former world record without losing its reactivity. As a result, we succeeded in direct observation of the reaction between [Fe(IV)(O)(Por·+)] and terminal C–H bond of an alkane substrate.
This work is a result of collaboration with Prof. Fujii in Nara Womans Univ.
ポルフィリンの一電子酸化体を配位子として持つ鉄四価オキシド錯体 [Fe(IV)(O)(Por·+)] (下の図、右)は、微生物から人間、植物に至るまで、幅広い生物の代謝過程における重要な化学種です。この化学種は活性が高すぎるがゆえ、反応溶媒ですら瞬時に酸化してしまいます。これまでの研究では、-60°Cといった極低温条件を用いることで、なんとかその観察を行っていました。
我々は、種々の溶媒を試した結果、トリフルオロトルエンを用いて実験を行うことによって、Fe(IV)(O)(Por·+) の分解が顕著に抑えられることを見出しました。これまでの最長記録と比べ、700倍以上も寿命が伸び、室温付近(30°C)でも、活性種を観測することに成功しました。
また、これまでのFe(IV)(O)(Por·+) についての研究では、溶媒の酸化が基質の酸化徒競走して進行してしまうがゆえに、基質は酸化されやすいものに限定されていました。溶媒の酸化を抑えることによって、実験に用いることができる基質が大きく広がりました。結果として、通常の反応では活性化することが困難な、アルカンの末端炭素のC–H結合を酸化することができるようになり、反応の様子を世界で初めて直接的に観測することに成功しました(下図、左)。
高い酸化活性を持つ化学種の反応機構は、分厚いカーテンに隠されていますが、それを開く方法が分かって来ました。この成果を手がかりに、より酸化の難しい物質を、より温和な条件で選択的に酸化する方法の発展が期待されます。この論文は、奈良女子大学藤井先生のグループとの共同研究の成果です。
31. Characterization and Reactivity of a Tetrahedral Copper(II)-Alkylperoxido Complex
Ikuma Shimizu; Yuma Morimoto; Gunasekaran Velmurugan; Tulika Gupta; Sayantan Paria; Takehiro Ohta; Hideki Sugimoto; Takashi Ogura; Peter Comba*; Shinobu Itoh*
Chem.–Eur. J. 2019, 25, 11157-11165 (DOI: 10.1002/chem.201902669)
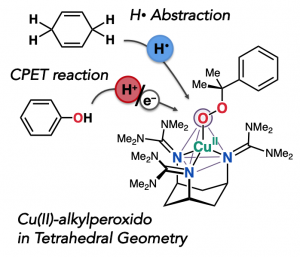
Generally, cupric complexes strongly prefer to have square or tetragonal pyramidal structure. In our group, we succeeded in the generation of Cu(II) complex in tetrahedral geometry by employing a ligand with rigid cyclohexane backbone and bulky guanidine moiety. Interestingly, some copper containing enzymes possess such Cu(II) reaction centers in tetrahedral geometries. As a follow-up work of IC paper published in 2017, we generated and characterized its alkylperoxido-Cu(II) complex. This complex abstracted hydrogen from carbon center, whereas its preferred to accept proton from oxygen center. Weak Cu-O bond may promote proton transfer than O–O bond cleavage. After dissociation of peroxide ligand, phenolate coordinated to the cupper center, followed by the reduction of cupper(II) to cupper(I). This work is result of collaboration with Prof. Comba (Heidelberg Univ) and Prof Ohta (Sanyo Onoda City Univ).
一般的に、四角錐あるいは平面四角形構造を好む銅(II)錯体ですが、我々のグループでは、固いシクロヘキサン骨格上に、嵩高いグアニジン部位をもたせた配位子を利用すると、四面体構造を有する銅(II)錯体が生成することを、以前報告しています(IC, 2017)。今回は、この錯体系を利用し、銅(II)-アルキルペルオキシド錯体を生成し、同定しました。よく見られるように、この錯体は炭素からの水素引き抜き反応を行った一方で、酸素からはプロトンを受け取ることが分かりました。おそらく銅-酸素結合が弱く、酸素-酸素結合の開裂よりも先にプロトン移動が起こるものと思われます。その後、フェノールアニオンにより、銅中心が還元されます。
酵素には、この錯体と類似の四面体構造の銅活性中心を有するものがあり、この研究は、銅の配位構造により酵素の反応性が繊細にコントロールされていることを示唆しています。ハイデルベルク大Comba先生と、山口東京理科大学太田先生との共同研究の成果です。
30. Cupric-superoxide complex that induces a catalytic aldol reaction-type C–C bond formation
Tsukasa Abe; Yuta Hori; Yoshihito Shiota; Takehiro Ohta; Yuma Morimoto; Hideki Sugimoto; Takashi Ogura; Kazunari Yoshizawa*; Shinobu Itoh*
Commun. Chem. 2019, 2, Article number: 12
On the paper described is an effect of ligand structure on the reactivity of cupper(II)-superoxide complex. A change in ligand side-arm by one methylene changes the character of superoxide species from electrophilic to nucleophilic, to give catalytic ability in aldol reaction.
銅錯体配位子のわずかな構造の違いが、その酸素錯体(スーペルオキソ錯体: 02[•–])の反応性に与える効果を明らかにした論文です。具体的には、先行研究では、メチレン鎖ひとつ分の違いによって銅–スーペルオキシド種の求核性が大きく高まり、アルドール反応を触媒するようになることが分かりました。九大吉澤先生、塩田先生のグループと、山口東京理科大学太田先生との共同研究の成果です。
2018
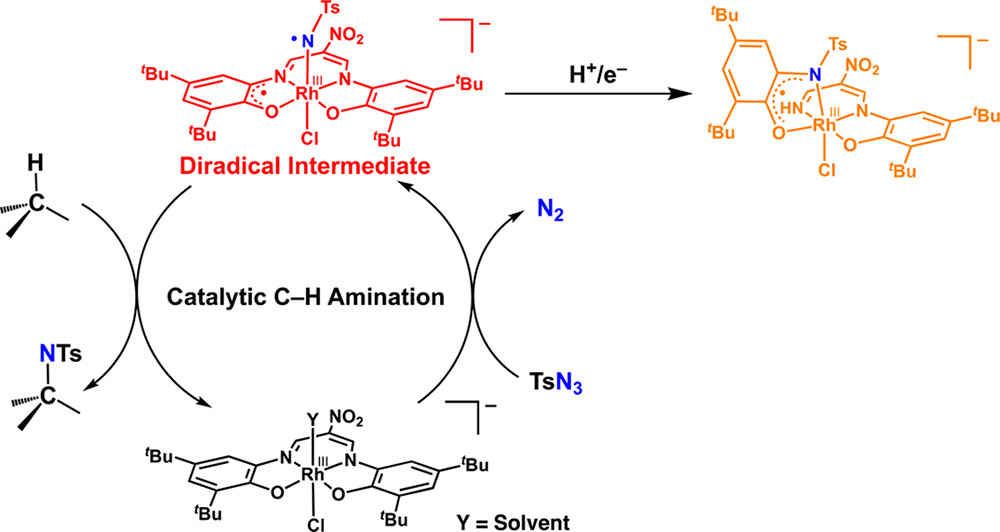
29. Non-innocent Ligand in Rhodium(III) Complex Catalyzed C-H Bond Amination with Tosyl Azide
Daiki Fujita; Hideki Sugimoto*; Yuma Morimoto; Shinobu Itoh*
Inorg. Chem. 2018, 57, 9738–9747.
This paper presents catalytic amination reaction of alkyl group with tosyl azide in the presence of Rh(III) catalyst supported by a ligand with redox ability (Redox-noninosent ligand). In the former report of rhodium complex with aniline ligands on the both axial positions, the very slow ligand exchange shackles the catalytic reaction. The presented rhodium complex becomes mono anion with chloride ligand on the axial position, and ligand exchange on the axial position was largely accelerated. From the reaction system, My fujita isolated another complex with a partially decomposed ligand with radical character.
トシルアジドを酸化剤&アミンソースとして、アルキル基のアミノ化を行った論文です。酸化還元活性を有する配位子を持つロジウム(III)錯体を触媒として用いています。前の報告では、ロジウム上のアミンがなかなか外れなかったのですが、軸配位子をクロライドとし、アニオン錯体としてやることによって、一般的には置換不活性なロジウムの配位子交換が加速されました。触媒反応系中から藤田くんは、配位子が部分的に開裂し、アミノフェノールのラジカル体となったものも単離してきました。DFTとEPRのパートを担当しました。
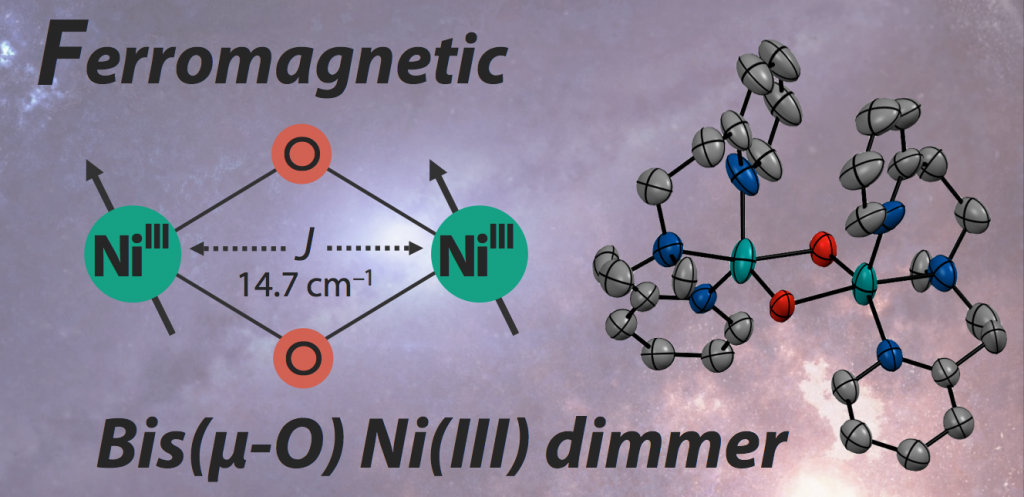
28. A Bis(µ-oxido)dinickel(III) Complex with a Triplet Ground State
Yuma Morimoto*; Yusaku Takagi; Toru Saito; Takehiro Ohta; Takashi Ogura; Norimitsu Tohnai; Motohiro Nakano; Shinobu Itoh*
Angew. Chem., Int. Ed. 2018, ASAP (DOI: 10.1002/anie.201802779)
Reported in this paper is a new crystal structure of nickel(III) di-nuclear complex showing variety of oxidation reactivities. In many case, spins on dimmer complex shows strong anti-ferromagnetic behavior in high-oxidation state with µ-oxide bridging ligand. This complex was found to have a triplet ground state due to the orthogonality between dz2 orbitals storing radicals. EPR and SQUID was employed in the discussion, strongly helped by DFT calculation.
酸化反応の活性種である、二核ニッケル(III)錯体の結晶構造と、そのスピン状態を決定した論文です。近年、酸化反応活性種の反応特性について、その電子状態から理解してゆこうという試みが進められています。その中で、電位や立体の因子に加え、スピン状態についても注目が集まっています。本、論文のニッケル錯体は非常に不安定な錯体でしたが、その結晶構造解析およびSQUIDやEPRを用いたキャラクタリゼーションに成功しました。その結果、高い酸化数を持つ金属酸素二核錯体としては極めて珍しく、強磁性的に相互作用した(スピンの向きが揃った)錯体であることがわかりました。電子相関の寄与を正確に見積もる必要のある錯体分子の計算は、一般には非常に難しいのですが、構造と電子状態のデータがしっかり取れていたので細かく議論することが出来ました。
27. Copper(I)–Dioxygen Reactivity in the Isolated Cavity of a Nanoscale Molecular Architecture
Sayantan Paria; Yuma Morimoto; Takehiro Ohta; Shinsuke Okabe; Hideki Sugimoto; Takashi Ogura; Shinobu Ito*
Eur. J. Inorg. Chem. 2018, 1976-1983. (DOI: 10.1002/ejic.201800029)
The cupper(I) complex has a supper bulky ligand stabilizing its reactive-oxygen complex. The complex shows temperature depending color change ascribed to the change in the binding mode of oxygen ligand. This possibility was validated by the comparison of their rRaman spectral bands and calculated values of their vibration modes.
銅(I)錯体と酸素分子の反応性に関する論文です。非常に嵩高い錯体の配位子が銅中心をぐるりと取り囲んでおり、中心の活性酸素錯体の崩壊を防いでいます。温度に依存した、各種スペクトルの変化から、銅と酸素の結合様式が平衡状態にあることを提案しています。非常に分子量の大きな錯体でしたが、銅(III)パーオキサイドと銅(II)スーパーオキシドについて構造最適化し、ラマンバンドのアサインに成功しました。

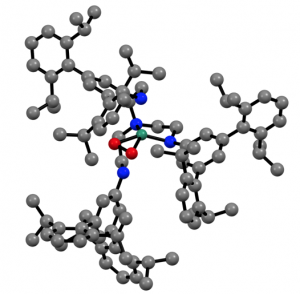
26. Structure and Reactivity of Copper Complexes Supported by a Bulky Tripodal N-4 Ligand: Copper(I)/Dioxygen Reactivity and Formation of a Hydroperoxide Copper(II) Complex
Sayantan Paria; Takehiro Ohta; Yuma Morimoto; Hideki Sugimoto; Takashi Ogura; Shinobu Itoh*
Z. Anorg. Allg. Chem. 2018, 644, 780–789 (DOI: 10.1002/zaac.201800083)
In this paper, we examined the reactivity of Cu(II)-OO• species toward hydrogen atom donor substrates employing a newly synthesized ligand TIPT2aped (shown below). Original bulky ligands (HIPT3tren and TIPT3tren) were too bulky to accommodate their substrates into the cavity.
この論文では、これまで報告してきたTIPT3trenリガンドの一部をピリジンに置き換えたTIPT2apedを用いて、銅(II)スーペルオキシド錯体の反応性についての検討結果を報告しています。スーペルオキシド配位子周りの空間に余裕が出ることで、大きく反応性が向上しました。
25. Tyrosinases in Organic Chemistry: A Versatile Tool for the α-Arylation of -Dicarbonyl Compounds
Roxanne Krug; Dennis Schröder; Jan Gebauer; Sanel Suljić; Yuma Morimoto; Nobutaka Fujieda; Shinobu Itoh; Jörg Pietruszka*
Eur. J. Org. Chem. 2018, 1789–1796 (DOI: 10.1002/ejoc.201800188)
Collaboration work with Pietruszka group in Universität Düsseldorf in Jülich, Germany
This paper is about tandem reaction of enzymatic oxidation reaction and organic addition reaction between keto-ester and quinone derivatives produced from aerobic oxidation reaction of phenol derivatives.
チロシナーゼにより、フェノール誘導体をオルトキノン誘導体へと空気酸化した後、系中に存在する求核剤と付加させる系を構築しました。これまで、ドイツのグループはラッカーゼ(カテコールをフェノールにする)を用いていましたが、伊東研究室で使っている麹菌(日本の国菌)由来のチロシナーゼを用いることで、カテコールよりも更に手に入りやすいフェノールを原料として用いることができるようになりました。酵素反応と有機反応のタンデムリアクションによって、酸化反応と四級炭素の構築反応が効率よく進行します。日本−ドイツの二国間共同研究支援事業で行った共同研究です。
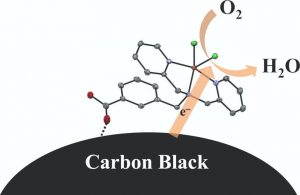
24. Crystal structure and oxygen reduction reaction (ORR) activity of copper(II) complexes of pyridylmethylamine ligands containing a carboxy group
Masafumi Asahi*; Shin-ichi Yamazaki; Yuma Morimoto; Shinobu Itoh; Tsutomu Ioroi
Inorg. Chim. Acta 2018, 471, 91-98 (DOI: 10.1016/j.ica.2017.10.031)
This is a paper for electrochatalitic oxygen reduction reaction with copper complex. Collaborative work with Dr. Asahi.
電極上に担持された銅錯体を用いた、酸素還元反応についての論文です。産総研の朝日さんのお仕事です。錯体に安息香酸部位を持たせてやることで、カーボンブラックの担体への担持効率が改善しました。
銅錯体のキャラクタリゼーション、特にEPRの部分を手伝いました。
2017
23. Geometric effects on O–O bond scission of copper(II)-alkylperoxide complexes
Tsukasa Abe; Yuma Morimoto; Kaoru Mieda; Hideki Sugimoto; Nobutaka Fujieda; Takashi Ogura; Shinobu Itoh*
J. Inorg. Biochem. 2017, ASAP (DOI: 10.1016/j.jinorgbio.2017.08.016)
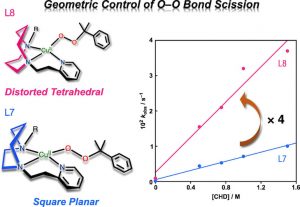
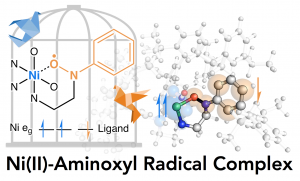
22. Generation and characterisation of a stable nickel(II)-aminoxyl radical complex
Arnaud Parrot; Yuma Morimoto; Sayantan Paria; Hideki Sugimoto; Nobutaka Fujieda; Shinobu Itoh*
Dalton Trans. 2017, 46, 8013-8016 (DOI: 10.1039/c7dt01789h)
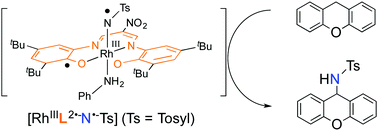
21. Catalytic C–H amination driven by intramolecular ligand-to-nitrene one-electron transfer through a rhodium(III) centre
Daiki Fujita; Hideki Sugimoto*; Yoshihito Shiota; Yuma Morimoto; Kazunari Yoshizawa*; Shinobu Itoh*
Chem. Commun. 2017, 53, 4849-4852 (DOI: 10.1039/c7cc01840a)
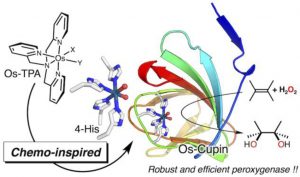
20. A Well-Defined Osmium–Cupin Complex: Hyperstable Artificial Osmium Peroxygenase
Nobutaka Fujieda*; Takumi Nakano; Yuki Taniguchi; Haruna Ichihashi; Hideki Sugimoto; Yuma Morimoto; Yosuke Nishikawa; Genji Kurisu; Shinobu Itoh*
J. Am. Chem. Soc. 2017, 139, 5149-5155 (DOI: 10.1021/jacs.7b00675)
19. Tetrahedral Copper(II) Complexes with a Labile Coordination Site Supported by a tris-Tetramethylguanidinato Ligand
Ikuma Shimizu; Yuma Morimoto; Dieter Faltermeier; Marion Kerscher; Sayantan Paria; Tsukasa Abe; Hideki Sugimoto; Nobutaka Fujieda; Kaori Asano; Takeyuki Suzuki; Peter Comba*; Shinobu Itoh*
Inorg. Chem. 2017, 56, 9634-9645 (DOI: 10.1021/acs.inorgchem.7b01154)
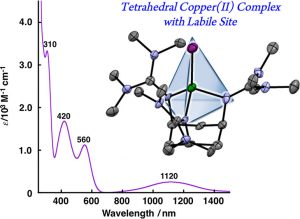
2016
18. Cerium-Complex-Catalyzed Oxidation of Arylmethanols under Atmospheric Pressure of Dioxygen and Its Mechanism through a Side-On μ-Peroxo Dicerium(IV) Complex
Mitali Paul; Satoru Shirase; Yuma Morimoto; Laurent Mathey; Balasubramanian Murugesapandian; Shinji Tanaka; Shinobu Itoh; Hayato Tsurugi; Kazushi Mashima
Chem. –Eur. J. 2016, 22, 4008–4014 (DOI: 10.1002/chem.201503846)
2015
17. Generation, Characterization, and Reactivity of a CuII–Alkylperoxide/Anilino Radical Complex: Insight into the O–O Bond Cleavage Mechanism
Sayantan Paria; Takehiro Ohta; Yuma Morimoto; Takashi Ogura; Hideki Sugimoto; Nobutaka Fujieda; Kei Goto; Kaori Asano; Takeyuki Suzuki; and Shinobu Itoh
J. Am. Chem. Soc. 2015, 137, 10870-10873 (DOI: 10.1021/jacs.5b04104)
16. Direct Hydroxylation of Benzene to Phenol Using Hydrogen Peroxide Catalyzed by Nickel Complexes Supported by Pyridylalkylamine Ligands
Yuma Morimoto; Shuji Bunno; Nobutaka Fujieda; Hideki Sugimoto; Shinobu Itoh
J. Am. Chem. Soc. 2015, 137, 5867-5870 (DOI: 10.1021/jacs.5b01814)
15. Redox Behavior of Novel Nickel and Palladium Complexes Supported by Trianionic Non-innocent Ligand Containing β-Diketiminate and Phenol Groups
Yuma Morimoto; June Takaichi; Shinichi Hanada; Kei Ohkubo; Hideki Sugimoto; Nobutaka Fujieda; Shunichi Fukuzumi; Shinobu Itoh
J. Porphyrins Phthalocyanines 2015, 19, 377-387 (DOI: 10.1142/S1088424615500248)
2014
14. A Tetradentate β-Diiminato Ligand Containing Phenolate Substituents: Flexivalent Coordination to MnIII, CoIII, NiII, and CuII
Fadhil Lafta Faraj; Hamid Khaledi; Yuma Morimoto; Shinobu Itoh; Marilyn M. Olmstead; Hapipah Mohd Ali
Eur. J. Inorg. Chem. 2014, 33, 5752-3759 (10.1002/ejic.201402745)
(University of Malaya, Malaysiaとの共同研究)
13. Geometric Control of Nuclearity in Copper(I)/Dioxygen Chemistry
Tsukasa Abe; Yuma Morimoto; Tetsuro Tano; Hideki Sugimoto; Nobutaka Fujieda; Shinobu Itoh
Inorg. Chem. 2014, 53, 8786 (DOI: 10.1021/ ic501461n)
12. Redox Chemistry of Nickel(II) Complexes Supported by a Series of Noninnocent β-Diketiminate Ligands
June Takaichi; Yuma Morimoto; Kei Ohkubo; Chizu Shimokawa; Takayuki Hojo; Seiji Mori; Haruyasu Asahara; Hideki Sugimoto; Nobutaka Fujieda; Nagatoshi Nishiwaki; Shunichi Fukuzumi; Shinobu Itoh
Inorg. Chem. 2014, 53, 6159 (DOI: 10.1021/ic5006693)
11. Unified View of Oxidative C−H Bond Cleavage and Sulfoxidation by a Nonheme Iron(IV)−Oxo Complex via Lewis Acid-Promoted Electron Transfer
Jiyun Park; Yuma Morimoto; Yong-min Lee; Wonwoo Nam; Shunichi Fukuzumi
Inorg. Chem. 2014, 53, 3618–3628 (DOI: 10.1021/ic403124u)
2013
10. Effects of Proton Acceptors on Formation of a Non-Heme Iron(IV)–Oxo Complex via Proton-Coupled Electron Transfer
Yusuke Nishida; Yuma Morimoto; Yong-Min Lee; Wonwoo Nam; Shunichi Fukuzumi
Inorg. Chem. 2013, 52, 3094–3101. (DOI: 10.1021/ic302573x)
9. Radical Chain Pathway in Formation of an Iron(IV)-Oxo Complex by Oxidation of an Iron(II) Complex with Dioxygen and Isopropanol
Yuma Morimoto; Yong-min Lee; Wonwoo Nam; Shunichi Fukuzumi
Chem. Commun. 2013, 49, 2500–2502. (DOI: 10.1039/c3cc38727e)
2012
8. Electron-Transfer Properties of a Nonheme Manganese(IV)-Oxo Complex Acting as a Stronger One-Electron Oxidant Than the Iron(IV)-Oxo Analogue
Heejung Yoon; Yuma Morimoto; Yong-Min Lee; Wonwoo Nam; Shunichi Fukuzumi
Chem. Commun. 2012, 48, 11187–11189. (DOI: 10.1039/c2cc36291k)
7. Mechanistic Borderline of One-Step Hydrogen Atom Transfer Versus Stepwise Sc3+-Coupled Electron Transfer from Benzyl Alcohol Derivatives to a Non-Heme Iron(IV)-Oxo Complex
Yuma Morimoto; Jiyun Park; Tomoyoshi Suenobu; Yong-min Lee; Wonwoo Nam; Shunichi Fukuzumi
Inorg. Chem. 2012, 51, 10025–10036. (DOI: 10.1021/ic3016723)
6. Proton-Promoted Oxygen Atom Transfer vs Proton-Coupled Electron Transfer of a Non-Heme Iron(IV)-Oxo Complex
Jiyun Park; Yuma Morimoto; Yong-Min Lee; Wonwoo Nam; Shunichi Fukuzumi
J. Am. Chem. Soc. 2012, 134, 3903–3911. (DOI: 10.1021/ja211641s)
2011
5. Scandium Ion-Enhanced Oxidative Dimerization and N-Demethylation of N,N-Dimethylanilines by a Non-Heme Iron(IV)-Oxo Complex
Jiyun Park; Yuma Morimoto; Yong-Min Lee; Youngmin You; Wonwoo Nam; Shunichi Fukuzumi
Inorg. Chem. 2011, 50, 11612–11622. (DOI: 10.1021/ic201545a)
4. Metal Ion Effect on the Switch of Mechanism from Direct Oxygen Transfer to Metal Ion-Coupled Electron Transfer in the Sulfoxidation of Thioanisoles by a Nonheme Iron(IV)-Oxo Complex
Jiyun Park; Yuma Morimoto; Yong-Min Lee; Wonwoo Nam; Shunichi Fukuzumi
J. Am. Chem. Soc. 2011, 133, 5236–5239. (DOI: 10.1021/ja200901n)
3. Metal Ion-Coupled Electron Transfer of a Nonheme Oxoiron(IV) Complex: Remarkable Enhancement of Electron-Transfer Rates by Sc3+
Yuma Morimoto; Hiroaki Kotani; Jiyun Park; Yong-Min Lee; Wonwoo Nam; Shunichi Fukuzumi
J. Am. Chem. Soc. 2011, 133, 403–405. (DOI: 10.1021/ja109056x)
2010
2. Dioxygen Activation by a Non-Heme Iron(II) Complex: Formation of an Iron(IV)-Oxo Complex Via C-H Activation by a Putative Iron(III)-Superoxo Species
Yong-Min Lee; Seungwoo Hong; Yuma Morimoto; Woonsup Shin; Shunichi Fukuzumi; Wonwoo Nam
J. Am. Chem. Soc. 2010, 132, 10668–10670. (DOI: 10.1021/ja103903c)
1. Crystal Structure of a Metal Ion-Bound Oxoiron(IV) Complex and Implications for Biological Electron Transfer
Shunichi Fukuzumi; Yuma Morimoto; Hiroaki Kotani; Panče Naumov; Yong-Min Lee; Wonwoo Nam
Nature Chem. 2010, 2, 756–759. (DOI: 10.1038/NCHEM.731)
Book
1. β-Diketiminates as Redox Non-innocent Supporting Ligands in Coordination Chemistry. in Chemical Science of π-Electron Systems
Shinobu Itoh; Yuma Morimoto
Springer Japan: 2015; pp 715–730
Perspective
2. 2-(2-Pyridyl)ethylamine (Pye) ligands in copper(I)-dioxygen chemistry
Shinobu Itoh; Tsukasa Abe; Yuma Morimoto; Hideki Sugimoto
Inorg. Chim. Acta 2017, ASAP. (DOI: 10.1016/j.ica.2017.09.017)
1. Mechanisms of metal ion-coupled electron transfer
Shunichi Fukuzumi; Kei Ohkubo; Yuma Morimoto
Phys. Chem. Chem. Phys. 2012, 14, 8472-8484. (DOI: 10.1039/C2CP40459A)
Commentary
2. 鉄と酸素が出会った瞬間をとらえる–ようやく姿を現したスーペルオキシド鉄錯体
森本祐麻, 伊東 忍
化学 2015, 70 (9), 64-65.
1. Aerobic Oxidation of Alcohols with TEMPO and Transition-metal Complexes
TEMPO と遷移金属錯体によるアルコールの空気酸化
森本祐麻
Bull. Jap. Soc. Coord. Chem. 2014, 63, 49-51. (DOI: 10.4019/bjscc.63.49)